Whenever a human is eating, a salt shaker is never too far away. For six thousand years we’ve relied on salt to improve our food. To make the uninteresting interesting and the bland, well, less bland. Though our ancestors often found it hard to get enough salt, in the modern world we are awash with the stuff. Without even seeming to try, the average human being on the planet consumes twice as much salt per day than what is considered healthy for us. Forget Dune and the spice of Arrakis, an alien stumbling across Earth would assume that its inhabitants were already addicted to their own crystalline spice.

But we can’t just stop eating salt. Going cold-turkey would certainly kill us. Salt is essential and without it our bodies would cease to function. Our muscles wouldn’t contract and our brain would lose touch with the rest of our body. Not that this would matter, our heart would also stop beating, removing the need for muscles or brains. Our bodies can’t make salt so to survive we rely on a constant supply of salt in our diet. But not too much. We need salt, but we need it like we need the movies of Adam Sandler, in just the right amount.
The root of our salt dependency begins with the cell. All cells, plant, animal and microbial, are bound by a membrane that marks the limit of the cell and presents a barrier to the outside world. This barrier, called a plasma membrane, is made of a lipid bilayer. I’ve discussed lipids elsewhere but for a quick refresher, lipids are a class of molecule that includes things that we would call fats, oils or waxes. One type of lipid is a fatty acid that has a charged, or polar, head group and a long hydrophobic (water-fearing) carbon chain. In a lipid bilayer two rows of fatty acids align with their hydrophobic chains interacting and their charged head groups facing outward. The plasma membrane is a continuous lipid bilayer that full encloses the cell, like a balloon.
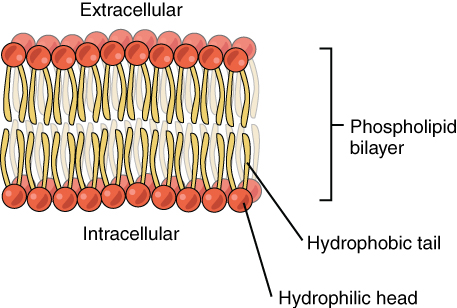
The arrangement of fatty acids in the plasma membrane is important. For a molecule to pass through a lipid bilayer it needs to be small and not carrying a charge. Small because of the tightness with which the lipids are packed and neutral because it needs to interact with, and pass through, all those hydrophobic tails inside the bilayer. A charged molecule doesn’t like interacting with hydrophobic molecules (as we saw in the emulsions post). This means many, if not most, organic molecules cannot spontaneously cross a lipid bilayer. They are too big, like some proteins, or they are charged, like sodium (Na+), or they are both, like sugars.
Importantly, water, being small and neutral, can cross the plasma membrane1. This makes the plasma membrane something scientists called a semi-permeable membrane. One of the characteristics of a semi-permeable membrane is that a solvent, usually water, will flow through the membrane from the side with fewer things dissolved in the water, things we call solutes, to the side with more solutes. You may remember this from high-school biology class, this process is called osmosis.
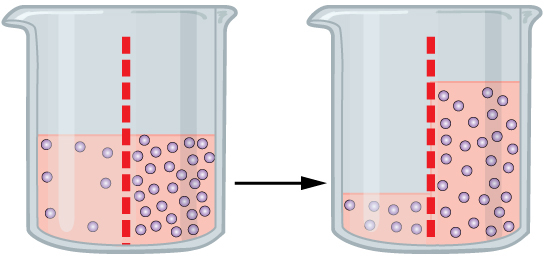
A cell, that is completely surrounded by a semi-permeable membrane, is very concerned with osmosis. Cells are packed with all sorts of molecules that it needs for its normal functioning. This means that a cell generally has more solutes within it than the surrounding water. This means that water will be constantly trying to enter the cell from the outside. This is good, cells need water, but when this flow of water is unregulated things can get difficult. If left unchecked the inflow of water can cause the cell to swell and, if things get real bad, it can even burst under this osmotic pressure. Conversely, if placed in a solution with a greater concentration of solutes it will shrink like a prune as water flows out of the cell.
There is nothing mechanical about osmosis, it’s a natural property of any membrane that allows the passage of water but not other molecules. This means that osmosis will occur whether the cell is alive or dead. In the kitchen you can see osmosis at work whenever you salt a steak or a pork chop. Water is drawn from the cells at the surface of the meat even though the cells are dead (you can check out my previous post on this, and its consequences for our BBQs, here). When we add salt, the high concentration of sodium (and chloride) on the exterior of the cells means there are more solutes on the outside than the inside and so the water flows out2 .
Salt is actually a good molecule if you want to adjust the osmotic potential across a membrane. It is a ionic compound with a positive sodium cation (a cation is a positive ion3) and a negative chloride anion (a negative ion). Salt is held together by the electrostatic attraction between the positive sodium and the negative chloride. But, when dissolved in water, it dissociates, which means it actually contributes twice as many solutes in a solution as it would if it didn’t dissociate. This is what makes salt so useful when brining meat, twice the osmotic pressure for the price of one.

This also makes salt very useful for animals looking to prevent their cells exploding from osmotic pressure4. Just like we use salt to promote the flow of water out of cells when preparing food, the body uses salt (and other electrolytes) to balance the osmotic pressure on cells by regulating the amount of salt that is dissolved in our blood and extracellular fluid. That is, by keeping the amount of solutes dissolved in the blood and extracellular fluids roughly the same as that as the interior of cells there will be no flow of water into or out of cells.
Now this sounds a lot easier than it actually is. If cells were only at the mercy of the sodium concentration in the blood they would be in a lot of trouble if we weren’t able to get salt or we became dehydrated for some reason. If you took a long jog you’d be dicing with death. Instead animal cells have evolved a system were they are able to manage the sodium concentration in their immediate environment. This gives them a buffer against fluctuating blood electrolyte levels.
All animal cells have a protein embedded in their plasma membrane that provides a passage for ions through the plasma membrane. This protein, called a sodium-potassium pump, does what the name suggests. It pumps sodium out of the cell and potassium into the cell. But it doesn’t do this in equal amounts, for every three sodium ions that the cell pumps out it pumps in two potassium ions. This has the effect of keeping the external sodium concentration high, which helps to counter balance the osmotic pressure that is generated by all the other molecules in the cell that it needs to keep functioning.
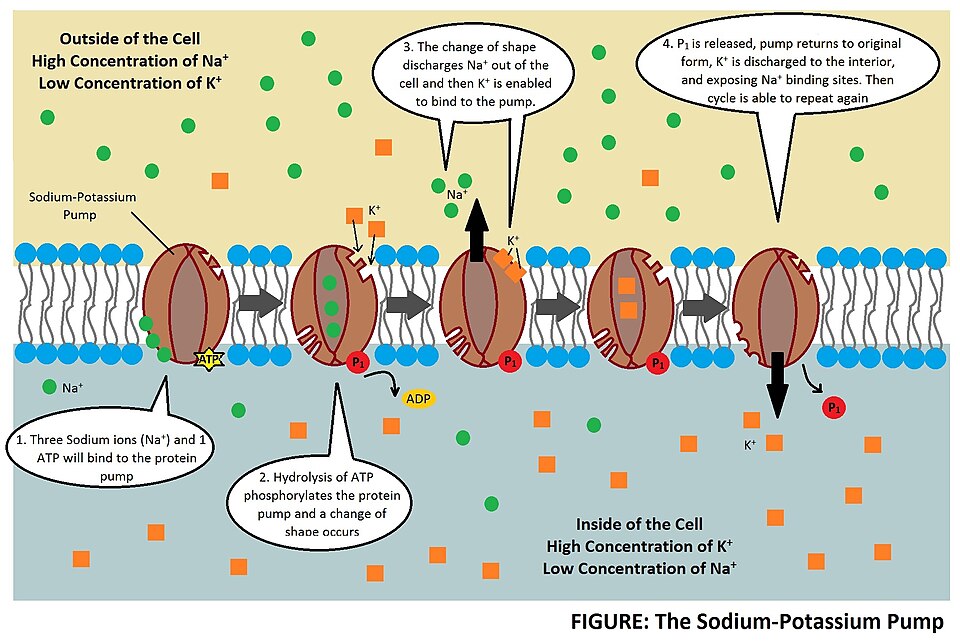
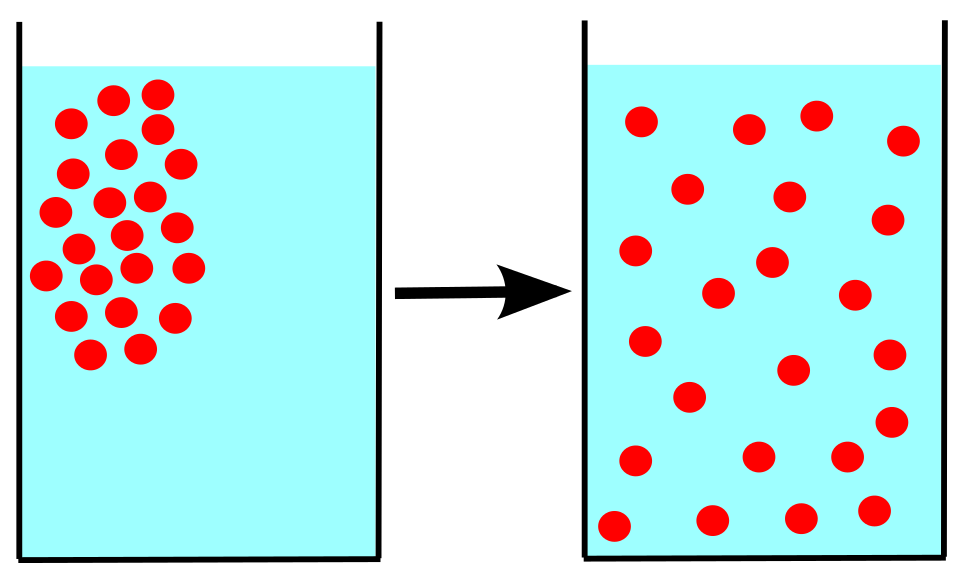
This is pretty clever of a cell, but cells have evolved to be far cleverer than that. Because there is now this sodium concentration gradient between the outside and inside of the cell, the cell can now use that gradient to move things it needs into the cell without expending any energy. Solutes naturally move down a concentration gradient. From a high concentration to a low concentration in the spontaneous process called diffusion5. The cell can utilise the energy of sodium diffusion by coupling the import of other molecules to the diffusion of sodium back into the cell.
Protein co-transporters, embedded in the plasma membrane, are the molecules that facilitate this coupling. There are transporters in cell membranes that drive the import of glucose, hydrogen, calcium and neurotransmitters into the cell by coupling their import to the diffusion of sodium. The magic (though it’s really physics) is that all this is achieved without the cell expending any energy. The cell does expend energy when using the sodium-potassium pump to establish the sodium gradient. But co-transporters allow the cell to avoid expending more energy importing other molecules against their own concentration gradient.
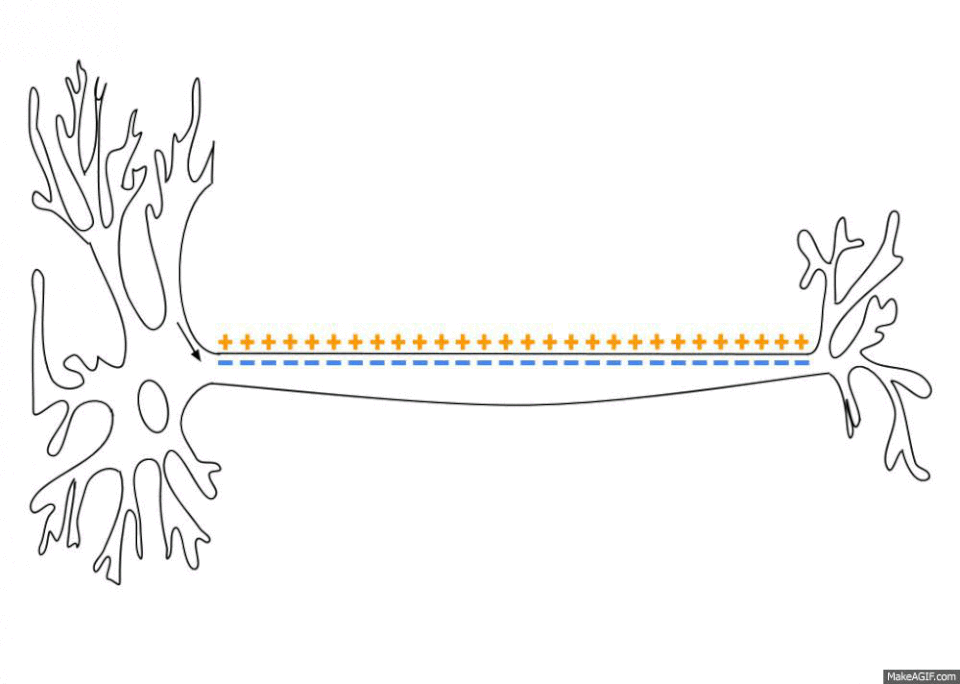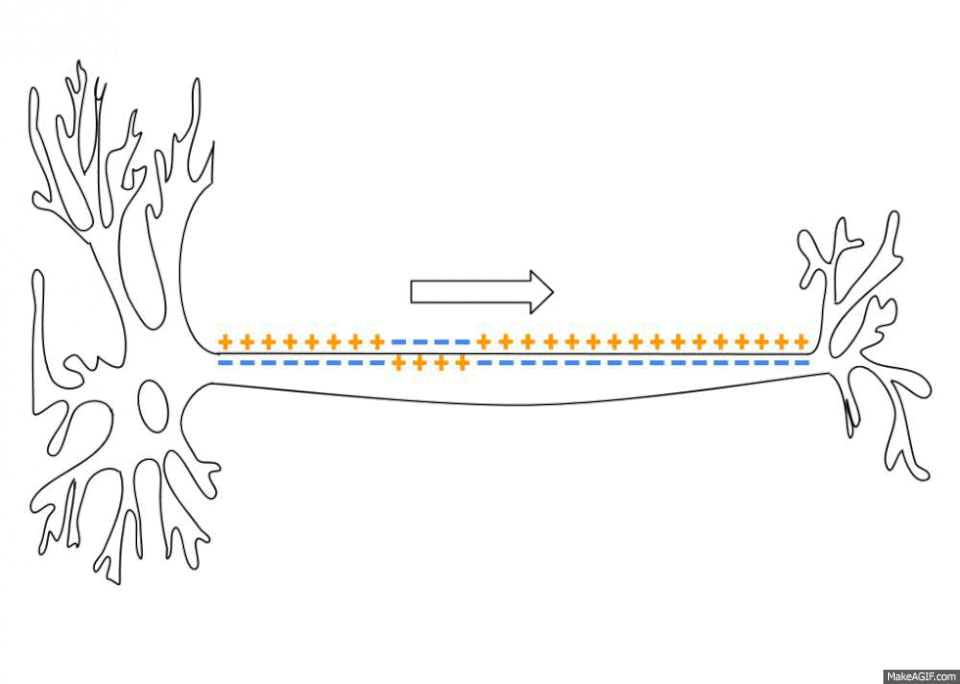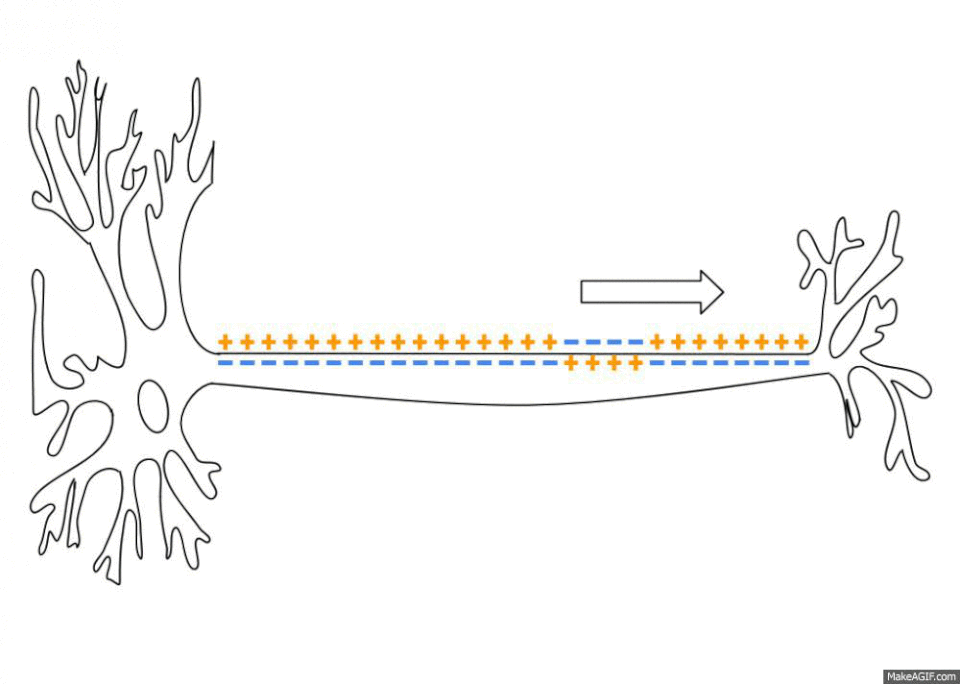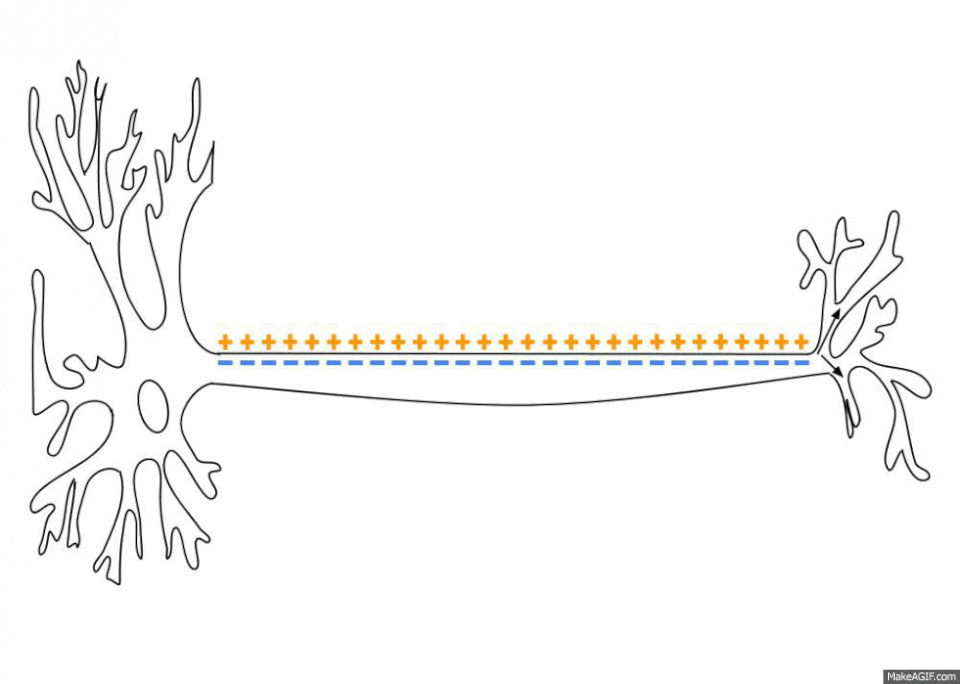
The sodium-potassium pumps haven’t stopped giving though. By pumping out three sodium ions (Na+) and importing only two potassium ions (K+) the sodium-potassium pump also establishes an electrochemical gradient across the plasma membrane6. This means that the inside of the cell is less charged than the outside of the cell, depending on the cell this difference can range from -20 to -200 millivolts. This is called the resting membrane potential and it is really important when you start looking at the propagation and transport of nervous signals as well as things like the contraction of skeletal muscles and the continued beating of your heart. There is no space to get into all this here but, no surprise, sodium plays a key role in all these processes.
So it seems that our cells have all this sorted without any concious effort from us. Cells balance some sodium, that’s pretty clever, but whats it got to do with my day to day life? Well, firstly, we get our sodium from salt and we lose some everyday in our urine, sweat, tears and, a tiny amount, in our faeces. This sodium needs to be replaced and that’s why a healthy adult needs around 2000 mg of sodium a day (which is around 5000 mg of salt, or about a teaspoon). This is not a problem for anybody in the modern age.
If think about it you can also see that sodium concentration can also fluctuate depending on how much fluid we have in our bodies. If you become dehydrated for some reason, say you are sick and vomiting a lot, you are also increasing the sodium concentration in your body. The less water in your body the more concentrated a given amount of sodium will be. If sodium concentrations become too high, a condition called hypernatremia, it can lead to cellular shrinkage, severe thirst, lethargy, and confusion.
Conversely, if you go on a long jog you will lose a lot of water but you also lose a lot of sodium and other electrolytes in your sweat. If you then drink a lot of water, without also replacing electrolytes, you are going to have a very low concentration of sodium and other electrolytes. When your sodium concentration is low, a condition called hyponatremia, you can experience things like confusion, severe headaches, dizziness, nausea, vomiting, and extreme muscle weakness. Drinking a lot of alcohol, a diuretic, can also cause lower our sodium levels, which might explain why we can get so hungry for fast food when we have a hangover.
So in day to day life our bodies are constantly trying to get us to behave in a way that maintains a healthy concentration of electrolytes in our body. Just like the regulation of hunger, our bodies regulate salt and water using a feedback system to maintain an appropriate water and salt balance.
Key players in this process are the brain, the heart and the kidneys. The kidney, in particular, are able to regulate salt and water loss by increasing water retention, stimulating thirst when we are dehydrated, reclaiming salts from urine when our salt levels are low or secreting electrolytes when electrolyte levels are high. A lot of what the kidney does, and how it communicates with the brain, involves the secretion of hormones like aldosterone (ALD) and angiotensin II (Ang II), that can stimulate salt appetite in the brain.
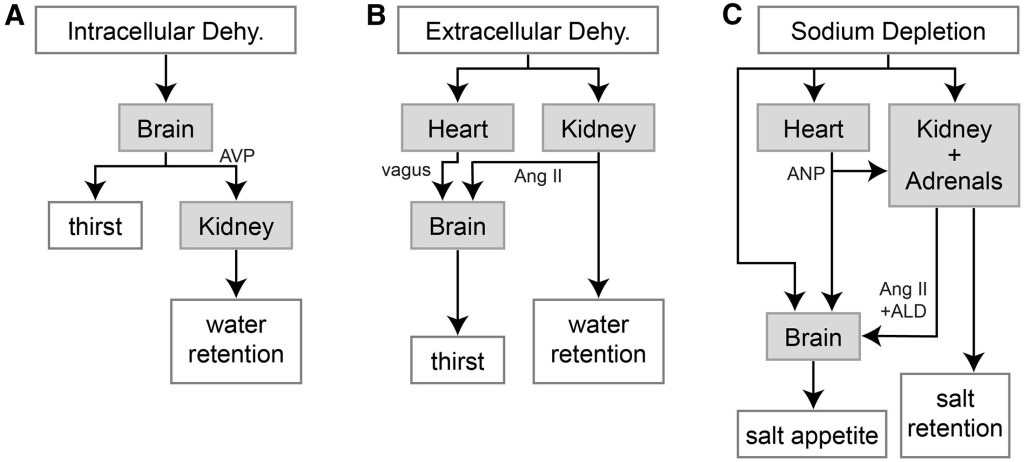
Mechanoreceptors in the heart are also able to sense dehydration. When we are dehydrated our blood becomes thicker, more viscous. When this happens the heart will communicate with the brain, via the vagus nerve, and the kidney, by releasing atrial natriuretic peptide (ANP), to stimulate thirst and water retention. Intracellular dehydration, when our cells don’t have enough water in their interior, can occur when there is an excess of salt in the blood. Remember, if there is an increase in the concentration of electrolytes outside the cell then water will flow out of cells. The brain can sense an increase in the osmolality of the blood, i.e. the number of solutes, and will make us get thirsty if it is too high. It will also release the hormone vasopressin (AVP) to stimulate water retention in the kidneys (if you want to dig deeper this is a good article).
We evolved this regulatory framework because for most of human history humans didn’t see a lot of salt. We get salt either from the evaporation of salty water, like the sea, or we mine it from large deposits left when ancient seas dried up. Especially, if you lived far from the sea and had no nearby salt deposits, it could be hard to get the amount of salt needed. As is so often the case in the modern world though, that has been flipped on its head and almost everyone on earth consumes far too much salt than they need. The global average daily intake of salt is more than double the 2000 mg recommended by the WHO.
The main culprit for this is processed foods that are often high in salt, but also sweet, so we don’t realise how much salt we are consuming. The body can store salt in various tissues around the body but this storage capacity can be exhausted and when that happens the sodium will circulate in the blood. We now know that high electrolyte concentrations are conceptually the same as being dehydrated. In these conditions the body will retain water to dilute the sodium and thus increase blood volume. Anytime you have more fluid going through the same pipes you increase the pressure and the human circulatory system is no different. High sodium intake causes high blood pressure and puts a strain on your heart that needs to deal with this increased pressure and keep blood moving around your body. The end result is often a heart attack.
We shouldn’t be beating ourselves up to much about this modern salt problem that we all share. We should be beating up food companies that over-sweeten and over-salt their processed foods. We can help ourselves by steering away from these types of food, but we pay the food companies for convenience and they should be able to provide convenience without a side helping of high blood pressure and heart disease. Even some clear labelling of highly salted foods would be a step in the right direction.
A classic example of this is the marketing of sports drinks. Portrayed as a healthy, hydrating alternative to water these drinks actually deliver a large amount of sodium and, often, sugar. It is true that anyone participating in intense and prolonged exercise needs to replenish electrolytes, but the absolute last thing that the average person needs is more sodium. We all like to entertain the idea that our jogs and trips to the gym are turning us into elite athletes. The truth is the vast majority of us don’t need to consume more electrolytes because we are getting more than twice the amount we need from our food.
Food companies have pushed all the responsibility for our diet onto us while creating the conditions in which it is hard to understand exactly what is in the food we are all being heavily encouraged to eat and drink. Until this changes we need to accept that responsibility but we should certainly be encouraging food companies to act a bit more responsibly any way that we can. In the meantime we need to understand why salt is important and how much we should be eating to keep ourselves and our families healthy. Because if we don’t do this for ourselves it’s highly unlikely that food companies are going to do it for us.
Footnotes
- It’s actually more complicated than this. Water isn’t charged but it is polar which slows down it’s diffusion across the membrane. But almost all living cells contain proteins embedded in the plasma membrane that allow the passage of water across the membrane. This greatly increases the rate at which water can flow in and out of the cell. ↩︎
- When it comes to meat, the cells are dead but they retain their structure, that is the plasma membrane is intact. When meats start to decompose that is when osmosis wont work anymore. The plasma membrane of the cells will have started disintegrating. ↩︎
- I actually remember this by remembering that the cat from Red Dwarf was cool and positive. Not the best mnemonic but it works. ↩︎
- I’ll mostly talk about animals here, plants avoid osmotic pressure by having a cell wall that allows them to withstand enormous amounts of osmotic pressure. This may have also doomed them to a stationary lifestyle. It’s hard to be mobile when every one of your cells need to be encased in armour like a medieval knight. ↩︎
- Think of a drop of ink in a glass of water. As time goes by the ink will fully diffuse through the water until there is a uniform concentration of ink through the water. Ultimately diffusion is the consequence of Brownian motion (the random thermal motion of particles) and Newton’s Second Law of Thermodynamics (the one about entropy) as described by Fick’s law. Good beginner article about it here. ↩︎
- Technically this is driven by passive potassium leak channels that leave the negatively charged proteins inside the cell. This is a dynamic process, potassium (K+) is forced out of the cell by its concentration gradient but pulled in by the negative interior. Calcium (Ca2+) and chloride (Cl–) are also important, particularly in muscles. But there are entire textbooks written on this and we’re probably getting too much in the weeds already. If you really want to get further into this topic there is a good resource here. ↩︎


Leave a comment