When we left vanilla in the last post, the French had finally broken the Spanish monopoly and were beginning to establish a vanilla industry in Madagascar and French Polynesia. Normally when a monopoly is broken prices come down as supply increases and the monopolist loses control over pricing. While this was partly the case with vanilla, you could really only say that prices went from eye-wateringly exorbitant to painfully expensive. Vanilla was, and still is, the most expensive spice after saffron and it was still mostly for the tables of the rich1.
The reason vanilla remained so expensive has a lot to do with the fact that it is an orchid. In fact vanilla is the only fruit of an orchid that we use as food. You can tell that vanilla is the fruit of an orchid by opening a vanilla pod. What you see are thousands of little seeds. This is typical of orchids because, unlike many other flowering plants, orchids do not rely on animals eating their fruit and distributing their seeds for them. They use the wind. Orchid fruit is not stuffed with fructose to make them attractive to passing mammals. Instead they have tiny seeds that, once the pods open, will disperse on the wind like dust.

Another peculiarity of orchids is that their seeds need to form a symbiotic relationship with a mycorrhizal fungi in order to germinate. The mycelial network of this type of fungus can form a relationship with plants in which they supply water and nutrients to the plant (I covered mycelial networks in my post on fungi). Distributing many thousands of their seeds by wind is a way that an orchid gives them the best chance of meeting a fungi that will help it germinate. Although vanilla seeds don’t have them, the seeds of many orchids have adopted to wind dispersal to the extent that they have a small “balloon” formed from the seed coat2 (if you want to learn more about orchid seeds this is a good article from Kew Gardens).

In vanilla production, of course, we don’t want seeds dispersing on the wind, we need them for our custard. But, if left to itself the vanilla fruit will split open longitudinally and release its seeds. Farmers need to pick the fruit before this happens but they also need to be patient. As soon as it is picked it will stop ripening and if it is picked too early what sugars the vanilla fruit does have will not have fully developed, impacting its eventual flavour. But picking the vanilla fruit is just the beginning, the reason vanilla costs so much is the months of work needed to transform fresh vanilla pods to the vanilla we all know and love.
If you take a green vanilla fruit and take a bite you won’t get a lot of vanilla flavour. It won’t kill you; it just won’t excite you that much. This is because a molecule that provides the most intense vanilla flavour, a molecule called vanillin, is conjugated with molecules of glucose in a fresh vanilla pod. Vanillin, when not conjugated to glucose, is very volatile, meaning that it readily evaporates and and can easily interact with receptors in our olfactory system (i.e. our nose and nasal cavity). This is where most of the “flavour” of vanilla comes from3. But, when conjugated to glucose it is significantly less volatile, meaning less of it will get into our olfactory system, reducing the amount of flavour that we perceive.

In the mouth, vanillin also interacts weakly with bitter receptors (see here for my post on our taste receptors), so if you eat it in high enough concentrations it will taste bitter. As we saw in my post on chillies, vanillin is also from the same class of molecules as capsaicin, the molecule that gives chillies their heat. Vanillin reacts with the same receptor in the mouth as capsaicin (the TRPV1 receptor), but it is either a very weak agonist (i.e. it only weakly activates the receptor) or, sometimes, a suppressor of the receptor (in which case it is acting as an antagonist). This is why custard, despite vanillins similarity to capsaicin, isn’t considered a spicy food. There could also be a practical application of this knowledge; if you’ve got a curry that has a bit too much heat, adding vanilla might make it less hot (see here and here)4.
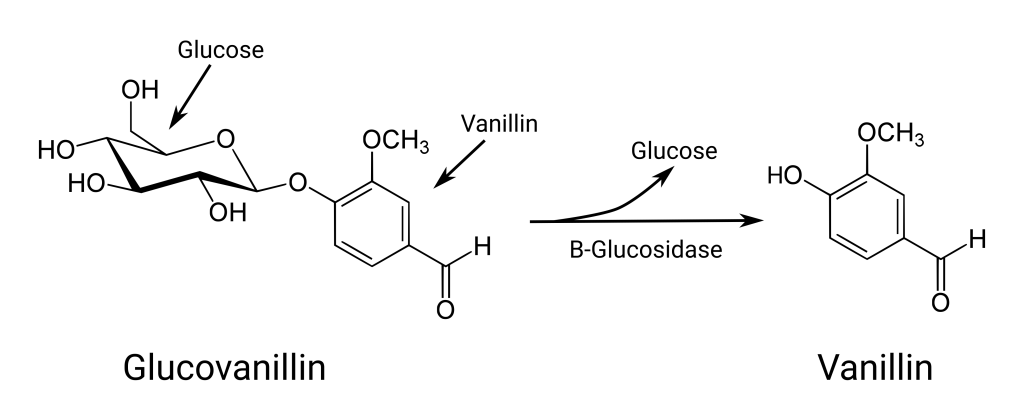
This vanillin chemistry is why picking the fresh vanilla pod is just the first step in a long curing process. Curing not only produces a well-dried and preserved vanilla pod, but it also gives time, and the right conditions, for the chemistry that unlocks the vanillin flavour from the fruit. Curing starts immediately after the fruit is picked. Firstly, the fruit is “killed”, a tricky process aimed at disrupting the cells of the fruit to stop further development and prevent the fruit splitting. This needs to be achieved without inactivating enzymes in the cells that will be important later in the curing process. Generally, this is achieved by dipping the fruit into 65C water for a specific period of time, though freezing the fresh pods also works.
After they are killed, and still warm, the fruit is wrapped in blankets and allowed to sweat for 1-2 weeks. This is where the vanillin flavour starts to develop as the conjugated vanillin is gradually released from its glucose molecule by enzymatic activity (just like the activation of glucosinolates in the Brassica that I covered a few weeks ago). 𝛽-Glucosidase, which hydrolyses the glucose from the vanillin, is the most important enzyme in this process though other enzymes are also important. For example, polyphenol oxidase breaks down phenols, and is the major contributor to the darkening of the vanilla pod, and other enzymes develop flavour molecules other than vanillin. Yet other enzymes break down cell walls and other cellular structures that will help with the drying process.

Finally, the pods are laid out to dry. This is yet another delicate process; the pods shouldn’t become too brittle, so you can’t just dump them in the sun all day, but you also want to control moisture and not let mold get a hold and destroy the whole crop. After a month or two the pods will have achieved the rubbery dryness that we associate with quality vanilla. A final step, called conditioning, normally gives the vanilla pods another two to four months of flavour development, though this can occur after packaging. All in all it can take six months to a year to get vanilla pods to market.
This whole process, which I’ve greatly simplified, is why vanilla has always been an expensive spice. You can’t just pick the fruit and send it off to market like you can do with apples and oranges. Vanilla needs some love and attention, and love and attention is always expensive.
At this point, given how hard to make and expensive vanilla is, you might be wondering why doesn’t vanilla ice-cream cost $100 a kilo? If you look at something like saffron, which is also hard to make, expensive but highly sort after, the world usage is something in the order of 200-300 metric tons per year. On the other hand, the world demands approximately 20,000 metric tons of vanillin per year for our ice-cream, custard and fragrances. Roughly one hundred times more vanillin is used per year than saffron (and that vanillin figure is from 2016). This level of vanillin consumption is far beyond what can be supplied by the vanilla plant and, by some estimates, less than 1% of the world’s demand for vanilla is met by agricultural production5.
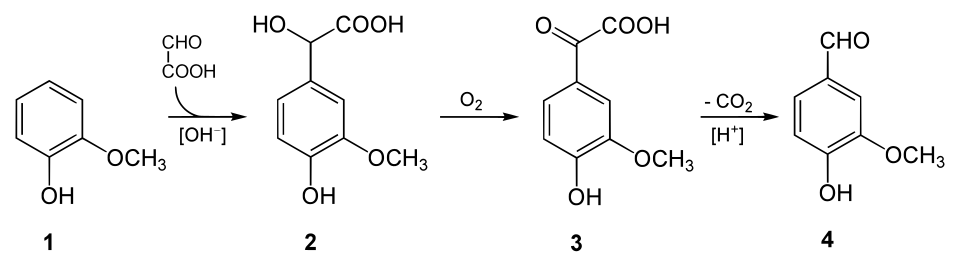
So, where is our vanillin coming from? The answer, of course, is that a whole lot of synthetic vanillin feeds our insatiable hunger for vanilla. Although vanilla was very popular almost from the instant it was introduced to Europe, in the 1520s, it took a while for chemistry to catch up and identify vanillin as the major flavour component. But chemistry did catch up; it was first identified in 1858, by Théodore Gobley, who recrystallised it from vanilla extract6. By 1874, German scientists Ferdinand Tiemann and Wilhelm Haarmann had started producing vanillin from pine bark and, later, from a molecule called eugenol found in clove oil.
Synthesis from clove oil remained the major source of synthetic vanillin until the 1930s when it was supplanted by a method that used lignin, a byproduct of the paper making industry. Environmental concerns eventually made lignin less attractive as a source of vanillin and today, almost 80% percent of our vanillin comes from, and I hate to say it, petrochemical sources. Guaiacol, a phenolic compound7, is a common molecule found in plants, but can also be formed in a multi-step process from benzene derived from petroleum. More recently, there’s been some interest in bio-vanillin, which is produced using microbes (see here if you want to know more about this).
Although vanillin produced through a synthetic process is exactly the same molecule that you’ll find in natural vanilla, it is important to realise that a vanilla pod has more than 200 other molecules that contribute to its flavour (see here for a comprehensive review). This complexity of flavour means that vanilla from different places, Madagascan versus Tahitian vanilla for example, will not only have different flavour profiles but that artificial vanilla, which only has vanillin, can’t really compete when it comes to flavour.
There is a caveat to this though. Although not necessarily rigorous science, a few different people have taste tested baked goods made with natural and synthetic vanilla. They mostly found that even experts couldn’t tell the difference. There is definitely a difference when tasting the pure product but it seems that the baking process might cook off most of the other flavour molecules, leaving vanillin which has a moderate resistance to heat (and maybe there is just more of it). It might also be the case that more subtle flavours of vanilla are overwhelmed by all the flavours going on in most baked goods. Either way, what this means for us is that we might get away using artificial vanilla in baked goods but, if we want to impress our dinner guests, we want real vanilla in things like custard or whipped cream where the vanilla is the star and the dish is prepared with less heat.
Although all the different ways we’ve developed to make vanillin is some pretty nifty science, to be honest, it almost seems hard not to produce vanillin as a byproduct of food production. Lignin, for example, is also the reason that we get vanilla notes in our wine, and other oak matured beverages like whisky. During their production oak barrels are heated and the heat causes production of vanillin from the lignin in the wood. When wines are matured in the barrel the vanillin leeches out of the wood and into the wine. American oak is better for this process than French oak because of it’s looser structure that seems to make it easier for the vanillin to leech out of the wood (there is a review of this topic here, though it’s getting a little old).

Likewise we find vanillin in roasted coffee, maple syrup, oatmeal, corn tortillas, olive oil, butter and some fermented foods, liked aged cheese. Sometimes this vanillin occurs naturally but often it is a result of the production process where heat converts plant molecules into vanillin. The chemistry of vanillin is the chemistry of phenols and plants are packed with phenolic compounds. It is a massive subject for another post but, here, it is enough to note that with all these phenolic compounds hanging around in plants it is little wonder that we see vanillin popping up every now and again, and thank god for that.
I’ve gone on way too long and I’ve barely scratched the surface of vanilla. Books could be written on the subject, and they have been, with some good recent examples here, here and here. Personally I am a big fan of vanilla; I love a buttery chardonnay and my favourite hangover cure is a large vanilla milkshake. A world without vanilla would be a very sad world indeed. I also have the feeling that the story of vanilla hasn’t been completely told yet, in 2019 a paper was published that showed evidence of vanilla usage in Mesopotamia 2,500 years before Cortez brought it back from the New World. How this is going to change our understanding of history I’m not sure but, I’m looking forward to finding out.
Footnotes
- There’s a good article on the pricing of vanilla here. ↩︎
- Though all this is true for most orchids there are some indications that vanilla is actually using it’s scent to attract animals for seed dispersal. You can read more about this here. ↩︎
- We perceive it as sweet because we have been trained to expect it in association with sweet dishes, something I discuss in my post on Michelin stars. ↩︎
- The association we have between vanilla and sweetness may also help with the heat, and adding vanilla might be better than adding sugar. I haven’t tried adding vanilla to a spicy curry, yet, but I will, if anyone else has tried it let me know. Maybe use artificial vanilla rather than the expensive stuff. ↩︎
- I’ve seen a bunch of different figures for this, ranging from 1% to 15% of the world’s supply coming from agricultural sources. I found the one percent figure in a couple of academic articles but I couldn’t trace it all the way back to a primary source. Might be one of those cases where everyone is citing each other. Either way a significant proportion of the worlds vanillin consumption does not come from the vanilla plant. ↩︎
- Vanilla extract is made by mashing up cured vanilla pods and percolating them in ethyl alcohol and water. This extracts vanillin and other flavour molecules from the pod. Ethyl alcohol is the colloquial name for ethanol, the same alcohol that is in your beer. ↩︎
- A phenolic compound is a large, diverse family of plant derived molecules that are characterised by at least one ring made of five or six carbons bearing one or more hydroxyl (-OH) group. In chemistry the rings are referred to as aromatic rings because they conform to Hückel’s rule (𝜋-electrons). OK I’ll stop the nerd talk but think of things like benzene and its derivatives: toluene, phenol, and aniline. ↩︎


Leave a comment